
Chapter 2: Simple Thermodynamic System:
2-1: Thermodynamic Equilibrium:
Thermodynamic coordinates change----->is called the change of state. For isolated system, this system is not influenced by its surroundings, which does not have much application to thermodynamics. So, when thermodynamic equilibrium reached, the system is equilibrium with its surroundings mechanically, chemically and thermally. Thus; thermodynamic equilibrium include the following conditions:
Mechanical equilibrium: no unbalanced forces between system and its surroundings.
Chemical equilibrium: no chemical reactions.
Thermal equilibrium: no temperature difference between system and surroundings.
States of thermodynamic equilibrium can be described in term of macroscopic coordinates that do not involve the time, i.e. in terms of thermodynamic coordinates.
(描述熱動力平衡系統的參數坐標是不隨時間改變的。)
"A system reaches thermodynamic equilibrium", that means, the system is in a state of thermodynamic equilibrium, and this state can be described in terms of macroscopic coordinates that do not involve in time, i.e. in terms of thermodynamic coordinates.
(The rest of our discussion concentrate on equilibrium state, not nonequilibrium case.)
Every system in thermodynamic equilibrium is a state which can be described by an equation of state (and the only one) which is specifically for "the state", though the equation of state may be so complicated that it cannot be expressed in terms of simple mathematical functions. For instance, ideal gas can be described by PV=nRT, but for real gas in the piston of car engine is not easy to be described by a simple mathematical form, also there exist so many parameters that it is not easy to find the relations among them. Therefore, an equation of state is not a theoretical deduction from thermodynamics but is usually an experimental addition to thermodynamics. Equation of state expresses the results of experiments in which the thermodynamic coordinates of the system were measured as accurately as possible, within a limited range of values.
Hydrostatic system: any system of constant mass (i.e. the total number of particles in the system will not exchange with its surroundings) that exerts on the surroundings a uniform hydrostatic pressure, in the absence of surface, gravitational, electric and magnetic effect.
Hydrostatic system can be
pure substance(由一種化學成份組成但可存在不同的相,phases),
Homogeneous mixture of different constituents(可由多種化學成份組成但存在於相同的相,phase),
Heterogeneous mixture(如氣液共存態).
Experiments show that the states of equilibrium of a hydrostatic system can be described with the aid of three coordinates, namely, the pressure P exerted by the system on the surroundings, the volume V, and the absolute temperature T.
2-2 PV Diagram For A Pure Substance:
Consider the system of a container about 2 cubic meters in volume from which all the air has been exhausted, and having 1kg of water at temperature 94oC introduced into it, thus the water will evaporate completely and the system will be in the condition know as unsaturated vapor, like point A. At point A, the system is compressed slowly and isothermally, the pressure will rise until there is saturated vapor at point B. If compression is continued, condensation will occur, but the pressure will not change ( this is an isobaric process). The line BC (vaporization line) represents the isothermal isobaric condensation of vapor.

在臨界點(液、氣共存點)時有一個特性,因為是一個 inflection point,所以下式成立;
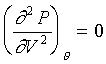
若在臨界點也是處於水平時→ ,如凡得瓦爾方程式。
,如凡得瓦爾方程式。
PV diagram 在低溫的部份(並未在上圖表出)有著固態相(solid phase),同樣地與液、氣之間類似的關係也存在固、氣相之間。也就是說會有一條類似BC的水平線,而這條線是液、氣的邊界同時也是固、氣的邊界。這條線上的點就是三相點(Triple
Point)。對一公斤的純水而言,在三相點時的壓力是611.2Pa,溫度是0.01oC,體積由10-3
m3(saturated liquid)到206 m3(saturated
vapor).
In Pq diagram of water, the slope of fusion curve (also called ice line) is negative because the volume become smaller when the ice melt, but for most material the slope is positive because the volume become lager when they melt. The slope of sublimation curve( for water, it can be called frost line) and vaporization curve (for water it can be called steam line) are always positive.
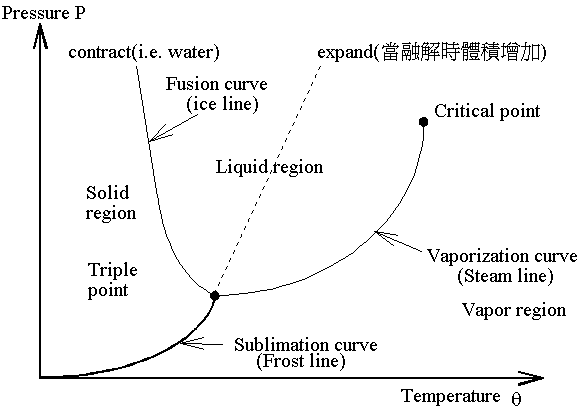
Triple point is the point of intersection of the sublimation, fusion and vaporization curves.
三相點在 Pq 圖中是一個點,但是在 PV 圖中是一條線。
冰在融解成水時,體積變小(而大多數物質則是膨脹),所以在上圖中Fusion curve的斜率是負值。即
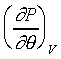 <0
<0
,表示在等體積情況下,壓力隨溫度的增加而變小。若是膨脹,則斜率是正值,即
 >0,
>0,
表示在等體積情況下,壓力隨溫度的增加而變大時,即融解成液體時,體積變大。
由 P, V, q 行成直角座標的相圖稱之為 PVq Surface。因為是一個面故稱之Surface。
註:在臨界溫度之上的部分稱之為 Gas phase,之下的部分稱之為 Vapor phase。
2-5 狀態方程式(Equations of State)
狀態方程式是指用數學式(某些可由理論導出絕大多數均為經驗式)描述每一個熱動力平衡態間P、V和q的關係,但是沒有一個數學式是可以用來描述整個相圖的範圍。單單在液態氣態(vapor)和液氣(liquid-vapor
region)的範圍,就有超過六十個公式,例如在低壓的蒸氣和氣體範圍可以用理想氣體方程式來描述
![]()
其中 v 表示每莫耳氣體的體積。另外在接近三相點附近,可以用有五個調整參數的
Beattie-Bridgman equation 來描述:(此式為一經驗公式)
![]()
其中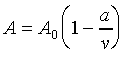
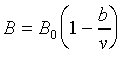 and
and ![]()
One of the most famous of the theoretical equations of states, based on assumptions concerning molecular behavior that are still of use today, is the van der Waals equation of state:

This equation holds fairly well in the liquid region, the vapor region, and near and above the critical point.
A, B and a, b, c are constant (or fitting parameters) in the above equations.
Home Work:
試以理想氣體方程式描繪 PV, Pq 和 PVq 的相圖。
試以凡得瓦爾氣體方程式描繪 PV, Pq 和 PVq
的相圖。
Every infinitesimal in thermodynamics must satisfy the requirement that it represents a change in a quantity which is small respect to the quantity itself and large in comparison with the effect produced by the behavior of a few molecules.
當系統由一個平衡態微小變化到另一個平衡態時,任何一個熱力學座標的微小變化、[一般以
dx (x 是熱力學座標) 來表示]或是任何熱力學測量量的微小變化,是指此的微小變化的大小與此測量整個值的大小來比要小的多,但是要比幾個分子對此測量量的影響要大的多。必竟熱力學是考慮巨觀的系統,如第一章所述。
現在讓我們來討論簡單的例子:如果體積可以表成壓力和溫度的函數,也就是
![]()

體膨脹係數(volume expansivity)可以寫成

等溫壓縮係數(isothermal compressibility)可以表示為
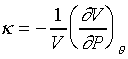
為什麼是負號呢!!是因為等溫壓縮係數是正值,壓縮時體積變小,所以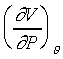 是負值。
是負值。
![]()
像上面這樣微分的方式(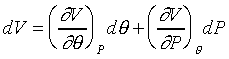 )是對一個確實的函數作微分,稱之為全微分(exact
differential)。若不是對一個確實的函數做微分則稱之為不全微分(inexact differential)。以數學方式來表示一個全微分是要符合下列條件,若x,y,z有下列函數關係
)是對一個確實的函數作微分,稱之為全微分(exact
differential)。若不是對一個確實的函數做微分則稱之為不全微分(inexact differential)。以數學方式來表示一個全微分是要符合下列條件,若x,y,z有下列函數關係
![]()
且此關係成立
![]()
則稱這微分為全微分。 全微分的外一個特性:若dV是全微分,則
![]()
上式表示全微分dV的積分與路徑無關。也就是因為如此,(在下一節中,會有下式成立)
若![]() 則
則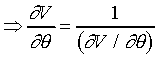
附加在β、κ上的一些論點: 實驗上証明對許多物質之β對壓力的變化並不敏感,且隨溫度的變化也很小,所以在很小的溫度範圍之內,β可以視為常數。 對於固體液體等溫壓縮係數κ,隨著溫度和壓力的改變,變化很小,故κ在此情況下常數,常被視為常數。 故在積分時,κ,β常被視為常數,而被提到積分外。
對理想氣體而言:![]()


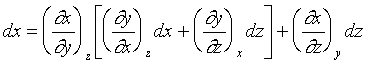
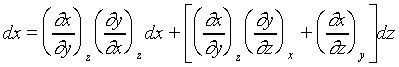


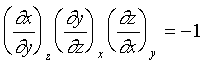
由上述的數學關係式;可以應用到靜流體系統。例如我們可以寫出:

或是

在上一節我們定義了體膨脹係數和等溫壓縮係數,
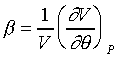

則β/κ的比值是:
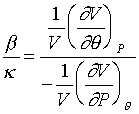
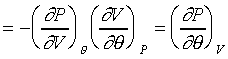
當壓力產生微小變化時,其可以寫成如下的形式:
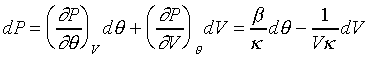
在體積保持定一定的時候:
![]()
此時溫度由初態溫度![]() 壓力
壓力![]() ,改變到未態溫度
,改變到未態溫度![]() 壓力
壓力![]() ,
則
,
則

以下用課本上的例子對上式作一個定量的計算:
A Mass of mercury at standard atmospheric Pressure and a temperature of 0oC is kept at constant volume. If the temperature is raised to 10oC, what will be the pressure?
因為 β=181×10-6 K-1 κ=3.82×10-11 Pa-1
(在此溫度範圍之內,β、κ被視為常數)
![]()
![]()
這種壓力是相當大的,這也就是為什麼溫度計過熱後會破裂,(若將量體溫度計置於50oC的熱水中,則此溫度計將失去功用。) 上面的討論是在等體積的情況之下,但是等壓力的情況又會如何呢?
| 例題:若有一溫度計是在等壓力的情況設計的,試比較其與等體積溫度計,何者在溫度計的刻度閱讀上較方便?(如果你是製造商,又會如何呢?) |
| Tension F
(N) Length L (M) Ideal-gas temperature θ, (Ko) |
在彈性限度之內,保持等溫的情況下
![]()
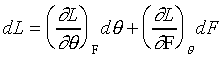
 (狀態方程式)
(狀態方程式)
其中 α 為線膨脹係數,等於  。A
為線的截面積,Y 為等溫楊氏係數(Isothermal Young's Modulus)等於
。A
為線的截面積,Y 為等溫楊氏係數(Isothermal Young's Modulus)等於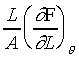 。
所以狀態方程式為
。
所以狀態方程式為
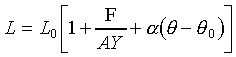 。
。
薄膜約可分為三種:
1.液體的最表層,其與表層外界的蒸氣 (vapor) 為持平衡。
2.肥皂膜或肥皂泡泡,其有兩個表面中間夾有液體。
3.油膜在水的表面(時是單分子層)。
先找此系統的熱力學座標
| 1.表面張力 N/m 2.薄膜面積 A m2 3.理想氣體溫度 q |
在討論表面薄膜時,壓力與體積並沒有被視為系統的熱力學座標。原因是壓力並不改變,而體積的變化可以被忽略。
實驗上證明,液體最表層薄膜的表面張力僅是溫度的函數。其狀態方程式為 =0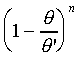 0 是在攝氏零度時的表面張力。q'表在臨界點附近的溫度,通常僅有幾度的差別。
油單層薄膜的狀態方程式是 (-w)A
= Const. q
w表潔靜水的表面張力,而表被油所覆蓋潔靜水的表面張力(因為油取代了部份的水)。(-w)稱之為表面壓力(Surface
Pressure)。
0 是在攝氏零度時的表面張力。q'表在臨界點附近的溫度,通常僅有幾度的差別。
油單層薄膜的狀態方程式是 (-w)A
= Const. q
w表潔靜水的表面張力,而表被油所覆蓋潔靜水的表面張力(因為油取代了部份的水)。(-w)稱之為表面壓力(Surface
Pressure)。
油膜在水面上或玻璃上有著彩色,為甚麼?
Pop Quiz: Removing stains: Some people when they get a greasy stain on their clothes, use a hot iron to remove it. What is the physics underlying this procedure?
Answer: The removal of greasy stains from clothes by ironing them is based on the fact that surface tension decreases as the temperature increases. So if a hot iron is applied to one side of the stained fabric and a piece of ordinary paper is pressed against the other side, the grease is transferred to the paper (or another piece of fabric that absorbs fatty substances)
一個系統的某些性質若與質量無關,則這些性質所代表的物理量是為強度量,如溫度、壓力、密度等。若某些性質與質量有關,則此量是為延伸量,如體積、能量等。簡單的測試方法是:將一個平衡系統等分成兩等分,質量也相等。單比較此兩系統中的物理量,均相等,但是在此小系統某些物理量變成原系統的一半,而有些量保持不變。保持不變的量稱之為強度量,而變成原來一半的量稱之為延伸量。